|
V-genes are grouped in families consisting of genes with at least 50% sequence homology. The high diversity of these chains is generated by stepwise recombinations of a multitude of variable ( V), in case of the β-chain diversity ( D), and joining ( J) gene sequences with a corresponding constant ( C) chain during thymic T-cell evolution. Recognition of Major Histocompatibility Complex class I (MHC-I) bound peptide is mediated by the dimeric transmembrane T-cell receptor (TCR) composed of an α- and a β-chain in the majority of cases. T-cell mediated immune responses are characterized by activation and subsequent clonal expansion of antigen-specific cells. T-lymphocytes are specialized mediators of the adaptive immune system, selectively destroying cells altered by viral infection or malignant transformation. The experimental strategy described allows the identification of alterations in the Vβ-family repertoire. We introduce a versatile method for the relative quantification of Vβ-families by real time PCR. The method was applied both to tissue and blood samples from patients with carcinomas and hematological disorders. We analyzed blood samples of 20 healthy volunteers for determination of mean and standard deviation for each family. Duplicates showed excellent technical reproducibility. Calculated percentages were in good correspondence with qRT PCR results demonstrating high reliability. The method was tested by serial dilutions of clonal T-cells in mononuclear cells from healthy volunteers. Total expression of α/ β TCR was assessed by quantification of the constant α-chain as a further control. 
Relative concentrations were calculated considering slopes and crossing points of each PCR reaction. Vβ-families were quantified using a universal reverse primer and family-specific forward primers with TaqMan technology on a light cycler instrument. Here, the heterogeneity of sequences within single families is the most challenging problem for exact quantification. Besides southern blotting and antibody-based approaches, quantitative real time PCR (qRT PCR) has been more widely applied in this context during the last years. These data provide supporting evidence for the hypothesis that acute GVHD is associated with expansion of T-cell clones expressing antigen-specific TCRs that may contribute to the disease pathology.Quantification of T-cell receptor (TCR) chain families can be utilized for detection of clonal T-cell populations. In most cases the probes are highly specific in detecting TCR expression from GVHD lesions alone, although in several instances expression could be detected in PB after GVHD diagnosis. Sequence-specific TCR CDR3 region probes were derived from analysis of the predominant expressed TCR in GVHD lesions, and used to probe Southern blots of amplified V beta 2 TCR mRNA from PB and tissue from BMT recipients and their respective donors. In contrast, GVHD-negative biopsy samples showed no evidence for clonotypic TCR amplification. We found evidence of oligoclonal expansion of single V beta 2-bearing TCRs in GVHD lesions, and in the PB of some patients after diagnosis of GVHD. V beta 2 TCR expression from these patients was analyzed more extensively using a combination of individual TCR gene cloning, followed by sequence analysis. In one such family, TCR V beta 2 is predominantly expressed in 7 of 7 biopsy specimens examined. Results of PCR studies from graft-versus-host disease (GVHD) skin lesions show a bias in the usage of TCR V beta families, whereas examination of peripheral blood (PB) withdrawn at the same time did not reveal a similar phenomenon. 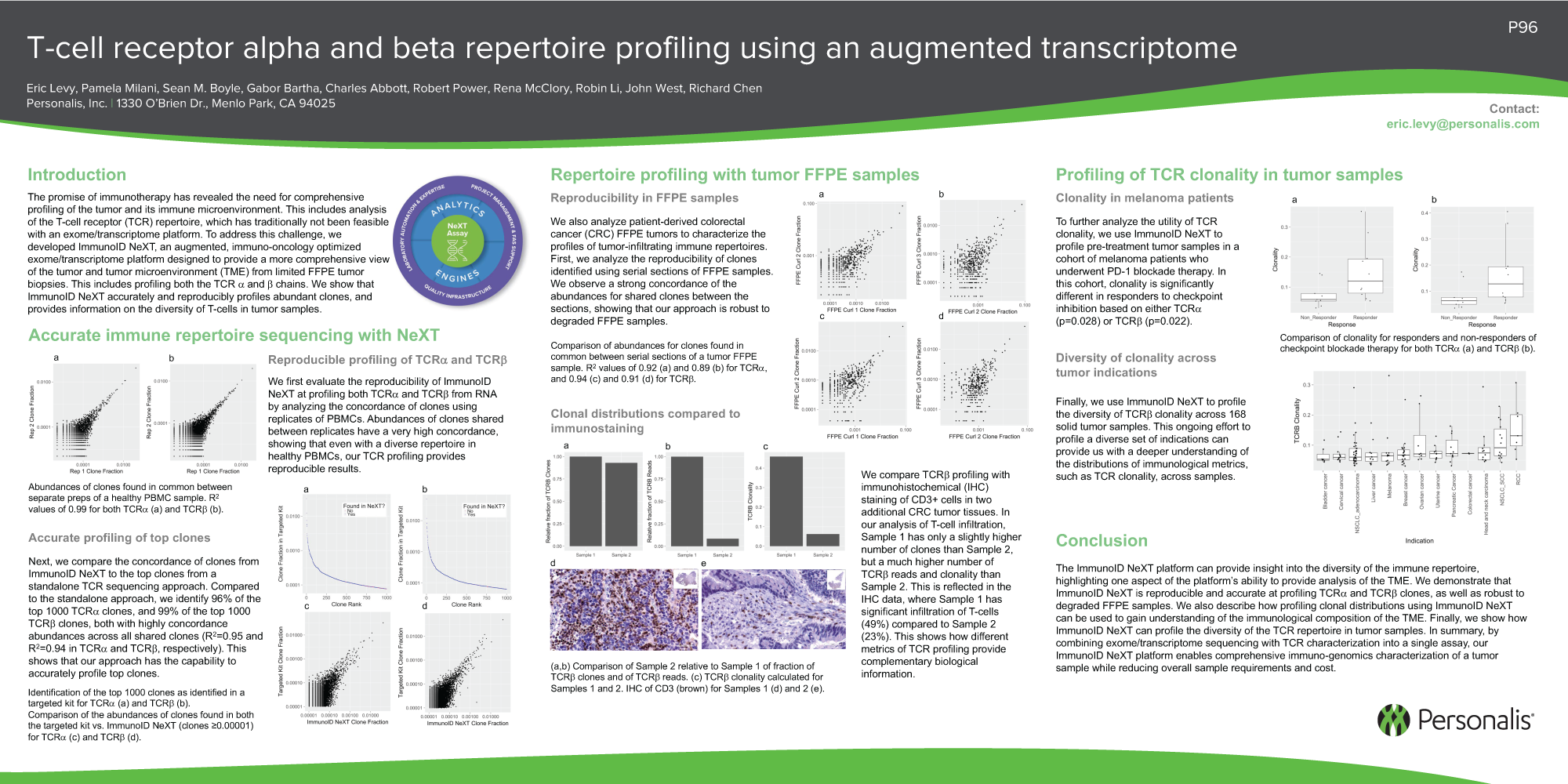 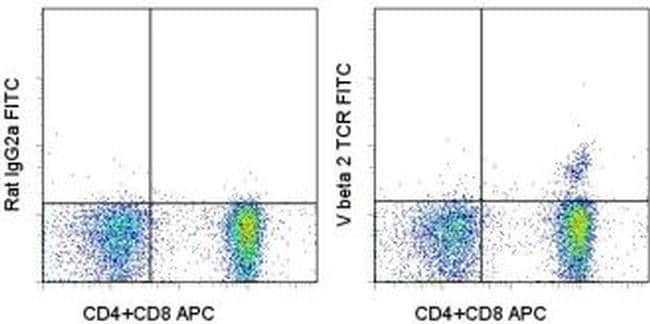
We have analyzed the T-cell receptor (TCR) V beta repertoire using polymerase chain reaction (PCR) in a cohort of eight patients receiving allogeneic bone marrow transplantation (BMT) from related and unrelated donors at the City of Hope.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
